ITQB NOVA research is “Paper of the Week”
Oeiras, 28.11.2016
Research by José Artur Brito, Inês Cardoso Pereira and Margarida Archer from ITQB NOVA, with co-workers from the University of Bonn (Germany), and the University of East Anglia (UK), has been selected as a Journal of Biological Chemistry Paper of the Week. The Editorial Board members and Associate Editors of the publication have determined that “Electron Accepting Units of the Diheme Cytochrome c TsdA, a Bifunctional Thiosulfate Dehydrogenase/Tetrathionate Reductase” is on the top 2% of manuscripts they will review in a year in significance and overall importance. Only about 50 to 100 papers are selected from the more than 6,600 that that journal publishes each year.
First authors José A. Brito, from ITQB NOVA, and Julia M. Kurth, from the University of Bonn, were highlighted by the Journal.
The sulfur cycle is a series of processes that enable sulfur to go to or from mineral sources to biological systems where it becomes part of essential molecules. Bacteria are key organisms in this cycle, as they comprise a series of enzymes that play a fundamental role in the association of sulfur in different chemical states.
The current work features the de novo crystal structure of the enzyme thiosulfate dehydrogenase from Marichromatium purpuratum, a purple sulfur bacteria isolated from marine sediments (from the latin, “the Chromatium from the sea dressed in purple”). Thiosulfate dehydrogenase enzymes are responsible for oxidation of thiosulfate to tetrathionate, or the reverse reduction of tetrathionate to thiosulfate.
In some bacteria, the diheme c-type cytochromes thiosulfate dehydrogenases, TsdA’s, interact with electron acceptor proteins named TsdB’s. Interestingly, in Marichromatium purpuratum, these two proteins are fused in a single polypeptide chain. Using spectrophotometric assays, enzymatic techniques and X-ray crystallography, this work shows how the interaction and electron transfer between TsdA and TsdB occurs at a molecular level.
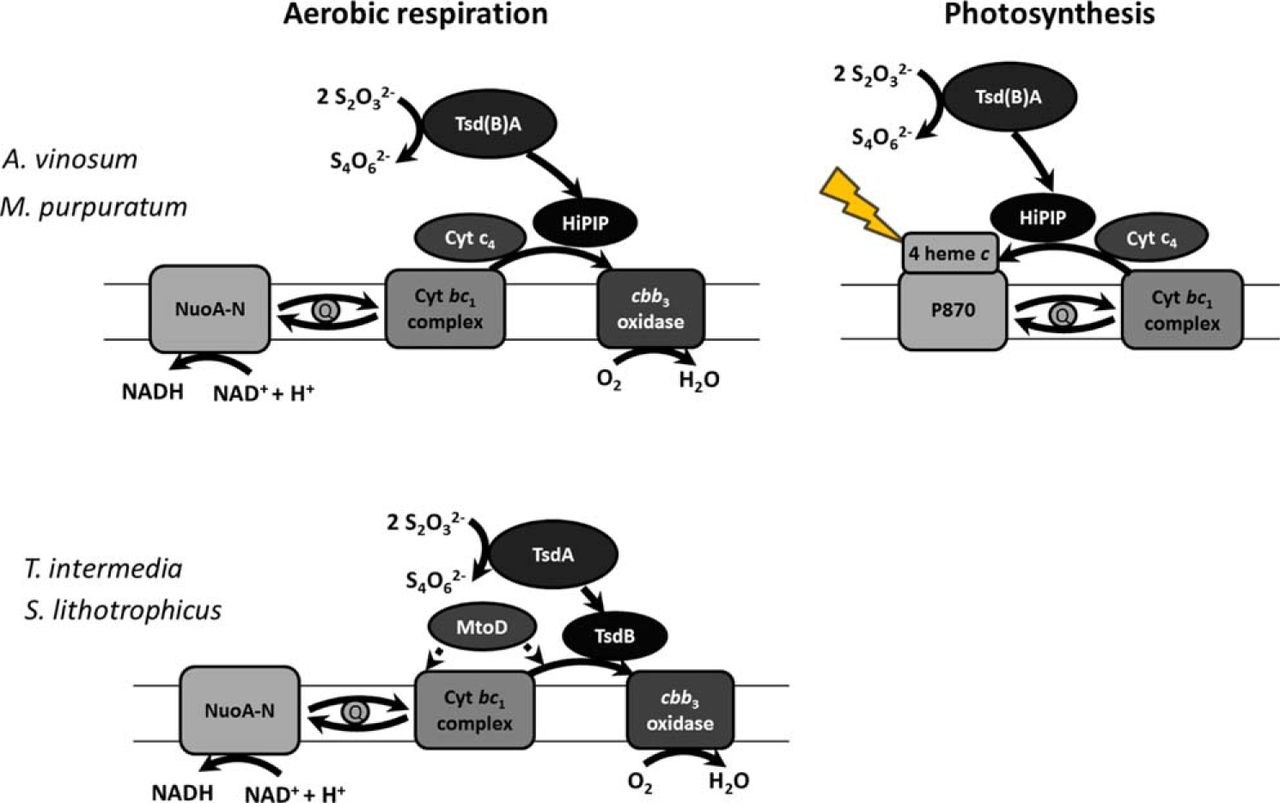
This work finally settles a controversy in the field that lasts for almost twenty years on the mechanism for thiosulfate oxidation, by showing that there is an intermediate state where thiosulfate is covalently bound to the enzyme. This intermediate could never be observed before.
ORIGINAL PAPER
J. Bio. Chem. 2016, 291, 24804-24818 DOI: 10.1074/jbc.M116.753863
Julia M. Kurth, José A. Brito, Jula Reuter, Alexander Flegler, Tobias Koch, Thomas Franke, Eva-Maria Klein, Sam F. Rowe, Julea N. Butt, Kevin Denkmann, Inês A.C. Pereira, Margarida Archer, Christiane Dahl







